На Македонски
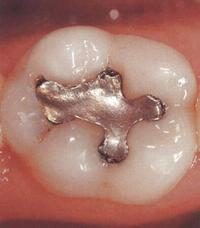
Dental amalgam is the oldest and the most useful filling material. To date, extensive research has failed to establish any links between amalgam use and general ill health.
Most dentists are continuing to use amalgam for cavities where a very durable material is needed - especially in the chewing surfaces of posterior teeth. In many respects amalgam is an ideal filling material. As well as being very durable, it expands and contracts with temperature change at the same rate as the surrounding natural tooth. Like most biomaterials, dental amalgam is not inert. Amalgam fillings release mercury vapor, especially when teeth are chewed on or brushed. Some of the vapor is exhaled but some reaches the rest of the body through inhalation. Part of it also dissolves in saliva and is swallowed. Much of the mercury entering the body is excreted but some accumulates in certain organs - especially the kidneys but also, to a very much lesser extent, in the brain, lungs, liver and gastrointestinal tract.
However, experts do not believe that the levels of mercury exposure resulting from amalgam fillings are of any general health significance. The amount of mercury that reaches the rest of the body is very small. Whether the amount is related to the number of fillings is unclear. But mercury is toxic, isn't it? Mercury is certainly toxic at large enough doses but the research does not indicate that the small exposures, which result from the use of dental amalgam, are harmful. Many substances are toxic at certain dose levels and for certain people, depending on individual susceptibility. Some people are even allergic to ingredients in foods such as bread and milk. Reactions sometimes occur in the soft tissues of the mouth next to fillings, not only with amalgam but with other restorative materials too. Amalgam is not especially allergenic and true sensitivity reactions are very rare. They may resolve spontaneously or after a change of restorative material. Suspected allergies are investigated by dermatologists/allergists and by oral medicine departments of dental hospitals, on referral from the dentist.
Should amalgam be used during pregnancy? It is known that mercury can cross the placenta from mother to foetus and can also be detected in breast milk but there is no evidence of any link between amalgam use and birth defects or still births. Generally, it is sensible to minimize health interventions during pregnancy. The 1998 report from the Committee on Toxicity already referred to say that there was no reason to think that the placement or removal of amalgam fillings during pregnancy was harmful. Committee on Toxicity agreed with the European Commission's expert group, however, that 'it may be prudent to avoid, where clinically reasonable, the placement or removal of amalgam fillings during pregnancy. More research was recommended. This does not constitute a ban on the use of dental amalgam during pregnancy. There will be occasions when patients will decide, on advice from a dentist, that the benefit of using amalgam (in terms of permanency, for example) outweigh the as yet theoretical risk of systemic toxicity. Dentists must always have a patient's informed consent before undertaking any treatment.
Should amalgam be used for children? Children who have a good diet and oral hygiene can usually be treated successfully using sealant resins and glass ionomer cements. But once posterior cavity approaches one third of the occlusal width and once any mesial or distal extension is not bound by enamel, glass ionomer fillings are not normally recommended and amalgam is an acceptable restorative material.
Is there any group for whom amalgam should not be used? Patients with proven amalgam sensitivity are the only group for whom the placement of new amalgam fillings is not advised.
Should dentists explain current concerns about amalgam safety when suggesting its use? Law and medical ethics require that patients be told enough about a proposed treatment and any associated material risks to enable them to reach an informed decision on whether to accept the treatment. The information given by the dentist will be a matter for personal judgement. In the case of a child with multiple allergies, the possibility of mercury or amalgam hypersensitivity might be raised, for example. However, based on recent legal decisions about the requirements for consent, dentists are advised that it is not necessary to discuss the alleged links between amalgam use and health problems with generally healthy patients, except in relation to pregnancy. Until there is a reputable body of opinion, which believes that there are material risks in amalgam use, consent given without discussion of side effects can be regarded as 'informed'. Dentists should, of course, always be prepared to answer patients' questions about the safety of dental amalgam.
Забниот амалгам е најстарото и најмасовно користеното средство за дефинитивно полнење на забните кавитети. Со денешните испитувања се бара врската помеѓу употребата на амалгамот и општата здравствена состојба. Повеќето стоматолози продолжуваат да го користат амалгамот, посебно за оние кавитети каде е потребно да се аплицира поцврст материјал, како што се џвакалните површини на моларите и премоларите. Во многу погледи амалгамот е идеално стедство за полнење. И покрај тоа што е цврст, неговата експанзија и контракција со промената на температурата е на исто ниво како и кај природните заби. Како повеќето био-материјали, забниот амалгам не е инертен. Амалгамските пломби испуштаат пареи на жива, особено кога се џвака или кога се мијат забите. Дел од тие пареи се исфрлаат со издишување. Дел се вдишуваат или се раствараат во плунката и се проголтуваат. Поголем дел од внесената жива се излачува од организмот, но еден мал дел се акумулира во органите, посебно во бубрезите, нешто помалку во мозокот, белите дробови, црниот дроб и гасрто-интестиналниот тракт.
Како и да е, експертите не веруваат дека количеството на жива кое е ослободено од амалгамските пломби има последици по здравјето на луѓето. Количеството на жива во организмот е многу мало, а дали тоа количество е сразмерно со бројот на амалгамски пломби е непознато. Сепак, живата во поголеми количества е отровна. Но, испитувањата покажуваат дека изложеноста на организмот на помали количества на жива, како при употреба на забниот амалгам е без поголеми последици по здравјето на човекот. Многу материи се отровни при одредени количества. Некои луѓе се осетливи на разни средства, како на пример поленот од цветовите, на млеко или млечни производи, а тоа зависи од нивната пречуствителност. Понекогаш, на слузокожата од усната празнина во близина на забната пломба може да се појави воспалителна реакција. Може да се работи за амалгамска пломба, но можеби во прашање е и некој друг материјал кој се применува во стоматологијата. Амалгамот не е алергенс и многу ретко може да се забележи вистинска алергична реакција. Алергијата може да се повлече спонтано или после отстранувањето на пломбата. Овие алергии се испитуваат од дерматолог/алерголог, како и на Клиниката за болести на уста.
Дали амалгамот треба да се користи за време на бременост? Познато е дека живата може да помине низ плацентата и да навлезе во фетусот. Живата може да се најде и во млекото од мајката, но не постои поврзаност помеѓу употребата на амалгамот и аномалии кај новороденчињата. Општо земено, разумно е да се минимизираат клинички интервенции за време на бременоста. Комитетот за токсикологија во1998 година изјави дека нема место да се мисли дека замената или отстранувањето на амалгамските пломби за време на бременоста е безопасна. Комитетот за токсикологија се согласи со Европската експертска група дека би требало да се одбегне замената или отстранувањето на амалгамските пломби за време на бременост онаму каде што е клинички прифатливо. Сепак, ова не е доволно да се забрани употребата на забниот амалгам за време на бременоста. Во такви случаи пациентите одлучуваат, со совет на стоматолог, кој им укажува дека користењето на амалгам може теоретски да предизвика систематско труење. Стоматологот мора секогаш да го информира својот пациент пред превземање на било какви зафати.
Дали амалгамот треба да се употребува кај децата? Доколку кавитетот кај постериотните заби е поголем од една третина кај децата, препорачливо е да се користат овие пломби, поради цврстината на амалгамот.
Кај која група на луѓе не би требало да се користи амалгамот? Пациентите кај кои е докажана пречуствителност од амалгамот се единствената група на луѓе кај кои употребата на амалгамски пломби не е препорачлива.
Дали стоматолозите терба да ги информираат своите пациенти за амалгамот кога ја препорачуваат наговата употреба? Според законот и медицинската етика во развиените земји се мисли дека пациентите се доволно информирани за да донесат сопствена одлука за предложениот третман. Информацијата дадена од терапевтот треба да послужи за донесување на таквата одлука. Во случај кога пациентот е алергичен на повеќе материи, можноста за пречуствителен на жива е злолемена. Како и да е, со донесени посебни одлуки се советуваат сите стоматолози дека не е потребно да се дискутира за врската меѓу употребата на амалгамот и здравствените проблеми кај релативно здравите луѓе.
Се до оној момент додека не постои доволен број на луѓе кај кои се видливи последиците од употребата на амалгамските пломби, не може да се донесе забрана за неково користење. Сепак стоматолозите треба да бидат подготвени да одговорат на сите прашање на пациентите во врска со опасностите кои ги носат овие пломби.
[ welcome
| what's new | student
exchange | congress
| articles | history
| the discussion list
| the people | contact
| help ]
© Macedonian Dentistry Students' Association
1999-2005